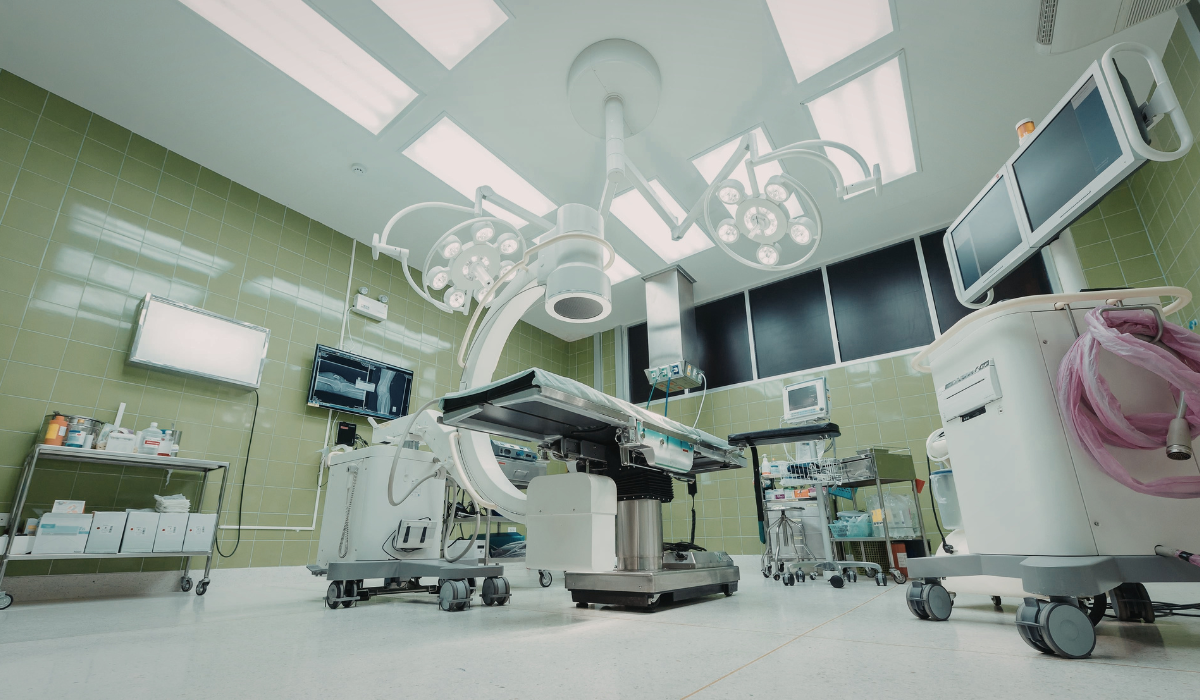
Cleanroom construction plays a critical role in helping medical device manufacturers meet strict regulatory requirements and maintain safe production environments.
Medical devices are used in healthcare settings where safety, precision, and reliability are essential, which means the manufacturing process must take place in controlled environments designed to minimize contamination.
In medical device production, even microscopic particles can affect product quality and patient safety.
Because of this, manufacturers rely on specialized facilities known as medical device cleanrooms that maintain strict environmental controls.
These facilities regulate airborne particles, temperature, humidity, and airflow to ensure that production processes remain consistent and compliant.
Many manufacturers partner with experienced cleanroom construction companies to build facilities that meet international standards and regulatory guidelines.
Through advanced engineering and facility design, custom ISO cleanrooms provide controlled environments that support compliance with industry standards while enabling efficient production processes.
Understanding how cleanroom construction supports compliance helps organizations maintain high manufacturing standards while protecting both product quality and patient safety.
Why Cleanrooms Are Essential in Medical Device Manufacturing
Cleanrooms play a critical role in medical device manufacturing because they create environments where contamination is carefully controlled.
Since medical devices must meet strict safety and quality standards, manufacturers rely on controlled production spaces that reduce airborne particles and maintain consistent environmental conditions.
These facilities help ensure that sensitive components are produced under conditions that protect product integrity while supporting regulatory compliance.
Protecting Product Safety and Quality
Medical devices often interact directly with the human body, which means they must be manufactured under carefully controlled conditions.
Even small contaminants such as dust particles, fibers, or microorganisms can compromise the integrity of a medical device.
Cleanrooms provide a controlled environment where airborne particles are limited to strict thresholds.
By controlling these variables, manufacturers can reduce contamination risks and ensure consistent product quality.
Supporting Regulatory Requirements
Medical device manufacturers must follow strict regulatory frameworks that govern production environments.
Cleanroom facilities are designed to support compliance with standards such as ISO cleanroom classifications and Good Manufacturing Practices (GMP).
Medical device cleanrooms allow organizations to maintain environmental conditions that meet these standards while ensuring that production processes remain controlled and traceable.
Without properly designed cleanrooms, manufacturers may struggle to maintain regulatory compliance.
Understanding Cleanroom Construction for Medical Device Facilities
Cleanroom facilities used in medical device manufacturing must be carefully planned and engineered to maintain strict environmental control.
The construction process involves more than simply building a controlled space.
It requires specialized design strategies that address contamination risks, regulatory standards, and operational efficiency.
Knowing how cleanroom construction works helps manufacturers develop environments that support both compliance requirements and reliable production processes.
What Cleanroom Construction Involves
Cleanroom construction involves designing and building environments that control airborne particles and environmental conditions.
These facilities require specialized engineering solutions, including filtration systems, airflow management, and contamination-resistant materials.
Cleanroom infrastructure must also support operational workflows, equipment placement, and maintenance procedures.
Proper planning ensures that the environment supports both regulatory compliance and efficient manufacturing operations.
Custom Facility Design
Medical device production processes vary widely depending on the type of device being manufactured.
Because of this, many facilities require custom ISO cleanrooms designed specifically for their operational requirements.
Cleanroom construction companies evaluate manufacturing workflows, contamination risks, and regulatory standards before designing facilities.
This customized approach ensures that cleanroom environments align with both production goals and compliance requirements.
Key Compliance Standards for Medical Device Cleanrooms
Medical device manufacturing is regulated by strict standards designed to protect product safety and patient health.
To meet these requirements, production environments must follow clearly defined guidelines that control contamination, maintain environmental stability, and ensure traceable processes.
Cleanroom facilities are designed to support these regulations by providing controlled environments that align with internationally recognized compliance standards.
ISO Cleanroom Standards
ISO cleanroom standards define the maximum allowable concentration of airborne particles in a controlled environment.
These standards categorize cleanrooms into different classes depending on the number and size of particles permitted in the air.
Manufacturers must maintain these classifications throughout production to ensure consistent product quality and regulatory compliance.
Good Manufacturing Practices (GMP)
Good Manufacturing Practices require manufacturers to maintain controlled environments, validated processes, and accurate documentation throughout the production cycle.
Cleanroom facilities play an important role in supporting GMP compliance by providing environments that reduce contamination risks and maintain controlled conditions during production.
Regulatory Expectations
Medical device manufacturers must demonstrate that their facilities meet regulatory requirements designed to protect patient safety.
This includes maintaining proper environmental controls, validating cleanroom performance, and documenting operational procedures.
Experienced cleanroom construction companies help organizations design facilities that align with these compliance expectations.
Core Elements of Medical Device Cleanroom Construction
Medical device cleanrooms are built using specialized engineering systems that help maintain strict environmental control.
These systems work together to regulate air quality, manage airflow patterns, and reduce the risk of contamination during production.
By integrating these critical elements into the facility design, cleanroom construction helps manufacturers maintain the controlled conditions required for safe and compliant medical device manufacturing.
Advanced Air Filtration Systems
High-efficiency particulate air (HEPA) filtration systems remove airborne particles from cleanroom environments.
These systems continuously filter air to maintain strict particle limits required for medical device manufacturing.
Controlled Airflow
Proper airflow design ensures that clean air flows through the facility in a controlled direction.
This prevents contaminants from entering critical production zones and maintains stable environmental conditions.
Cleanroom-Compatible Materials
Cleanroom construction requires materials that resist contamination and support frequent cleaning.
Surfaces must be smooth, durable, and resistant to chemical disinfectants used in sanitation procedures.
Environmental Monitoring Systems
Monitoring systems continuously track conditions such as temperature, humidity, and airborne particle levels.
These systems help manufacturers maintain compliance and detect environmental changes that could affect production.
Together, these elements support the reliable operation of medical device cleanrooms used in manufacturing environments.
How Custom ISO Cleanrooms Improve Manufacturing Efficiency
Every medical device manufacturing facility has unique production workflows and equipment requirements.
This is why many manufacturers invest in custom ISO cleanrooms designed to support their specific operations.
Customized cleanrooms can improve workflow efficiency by organizing production zones according to manufacturing stages.
This helps minimize contamination risks while maintaining efficient movement of materials and personnel.
In addition to improving contamination control, custom cleanroom designs can support future expansion and technology upgrades.
By integrating scalable infrastructure during cleanroom construction, manufacturers can adapt to changing industry requirements and evolving regulatory standards.
The Role of Cleanroom Construction Companies in Regulatory Compliance
Building a compliant medical device manufacturing environment requires more than construction expertise.
It involves a structured process that ensures every stage of the project aligns with regulatory requirements and operational goals.
Experienced cleanroom construction companies guide organizations through planning, design, construction, and validation to ensure that the final facility meets industry standards and supports reliable manufacturing operations.
Planning and Consultation
Professional cleanroom construction companies begin projects by evaluating operational needs, production processes, and regulatory requirements.
This ensures that facility designs align with industry standards.
Design and Engineering
Cleanroom engineers develop layouts that optimize airflow, equipment placement, and personnel movement.
Proper design reduces contamination risks while supporting efficient operations.
Construction and Installation
During construction, specialized materials and engineering systems are installed to maintain environmental control.
Each component must meet strict performance standards to ensure compliance.
Validation and Testing
After construction is complete, facilities undergo validation procedures to confirm that environmental conditions meet required ISO classifications and operational standards.
Through this process, cleanroom construction companies help organizations build facilities that support safe and compliant manufacturing operations.
Long-Term Benefits of Proper Cleanroom Construction
Investing in well-designed cleanroom facilities provides long-term benefits for medical device manufacturers.
These benefits include improved product quality, reduced contamination risks, and greater operational reliability.
Cleanrooms also support regulatory inspections by providing controlled environments that meet compliance requirements.
When organizations invest in advanced cleanroom construction, they create manufacturing environments that support consistent production performance and regulatory approval.
Common Challenges in Medical Device Cleanroom Projects
Medical device manufacturers often face several challenges when planning cleanroom projects.
These challenges may include adapting to evolving regulatory requirements, managing contamination risks, and optimizing facility layouts.
Scaling manufacturing operations can also create additional complexities.
As production demand increases, facilities may need to expand or reconfigure cleanroom spaces.
Experienced cleanroom construction companies help address these challenges by providing customized design solutions and scalable facility planning strategies.
Why Partnering With Cleanroom Construction Experts Matters
Medical device manufacturing environments require specialized knowledge of contamination control, engineering systems, and regulatory standards.
Partnering with experienced professionals helps organizations design facilities that meet both operational and compliance requirements.
Through strategic planning, advanced engineering, and specialized construction methods, cleanroom construction companies help manufacturers create environments that support reliable production processes.
With the right expertise, organizations can build facilities that maintain strict environmental controls while supporting efficient manufacturing operations.
Cleanroom Construction as the Foundation of Medical Device Compliance
Cleanroom environments are essential for maintaining safety and regulatory compliance in medical device manufacturing.
From controlling airborne particles to maintaining consistent environmental conditions, cleanroom facilities play a central role in protecting product quality.
Through careful planning and engineering, cleanroom construction enables manufacturers to meet regulatory requirements while maintaining efficient production workflows.
Custom facility designs also allow organizations to adapt their operations as technology and industry standards evolve.
By investing in properly designed custom ISO cleanrooms, manufacturers create controlled environments that support long-term compliance, operational reliability, and product safety.
Cleanroom Solutions for Medical Device Manufacturing
At UltraPure Technology, we are expert in designing and building advanced cleanroom environments that support regulatory compliance and operational efficiency.
Our team works closely with manufacturers to develop custom ISO cleanrooms tailored to the unique requirements of medical device production.
As experienced cleanroom professionals, we understand the importance of precise environmental control, reliable infrastructure, and scalable facility design.
Our expertise in cleanroom construction allows us to help organizations create manufacturing environments that meet strict industry standards.
If your organization is planning a cleanroom project or expanding a medical device facility, you can contact us. Our team can help design controlled environments that support compliance and long-term manufacturing success.
Frequently Asked Questions
Why is cleanroom construction important for medical device manufacturing?
Cleanroom construction creates controlled environments that reduce airborne particles and contamination risks during medical device production. These facilities help manufacturers maintain consistent environmental conditions that support product safety and regulatory compliance. By controlling airflow, filtration, and environmental monitoring, cleanrooms help ensure reliable manufacturing processes.
What are custom ISO cleanrooms?
Custom ISO cleanrooms are specialized controlled environments designed to meet specific manufacturing requirements while maintaining ISO cleanroom classifications. These cleanrooms are tailored to production workflows, equipment placement, and contamination control needs. Custom designs allow manufacturers to maintain compliance while optimizing operational efficiency.
How do cleanroom construction companies support regulatory compliance?
Cleanroom construction companies design and build facilities that align with industry regulations such as ISO cleanroom standards and Good Manufacturing Practices. They evaluate production requirements, engineer contamination control systems, and validate facility performance to ensure regulatory compliance.
What standards apply to medical device cleanrooms?
Medical device cleanrooms typically follow ISO cleanroom classifications and Good Manufacturing Practices guidelines. These standards define particle limits, environmental monitoring requirements, and operational protocols. Maintaining these standards helps manufacturers produce safe and reliable medical devices.
What industries require medical device cleanrooms?
Medical device cleanrooms are used in industries that manufacture products intended for healthcare use. These environments are essential for producing devices such as surgical instruments, diagnostic equipment, implants, and sterile packaging materials while maintaining strict contamination control.
How do cleanrooms reduce contamination in manufacturing?
Cleanrooms use advanced filtration systems, controlled airflow, and contamination-resistant materials to reduce airborne particles. Environmental monitoring systems track conditions to ensure that manufacturing environments remain within acceptable limits throughout production.
What factors influence cleanroom construction design?
Cleanroom construction design depends on production workflows, regulatory standards, equipment requirements, and contamination risks. Engineers evaluate these factors to create facility layouts that maintain controlled environments while supporting efficient manufacturing processes.
How long does a cleanroom construction project typically take?
The timeline for cleanroom construction varies depending on facility size, complexity, and regulatory requirements. Planning, design, construction, and validation phases must all be completed to ensure the facility meets performance and compliance standards.
Why do manufacturers choose custom cleanroom solutions?
Manufacturers choose custom cleanroom solutions because different production processes require unique environmental controls and facility layouts. Custom cleanrooms allow organizations to design environments that support their operational needs while maintaining compliance with industry standards.